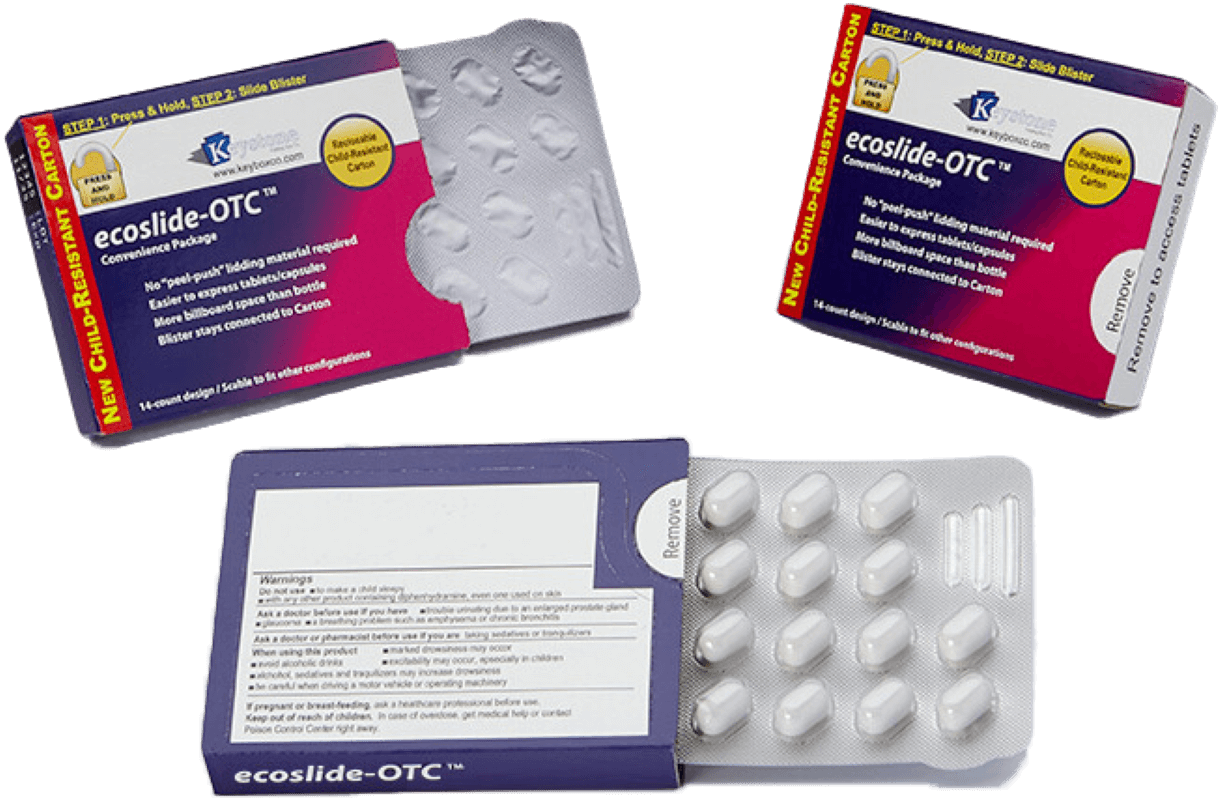
CR Packaging for Pharmaceutical Products
Keystone Folding Box Co. designs and manufactures innovative Child-Resistant (CR) packaging for pharmaceutical blister products, helping drug manufacturers meet critical safety regulations while enhancing the patient experience. Our patented paperboard-based platforms are engineered to prevent accidental child access and ensure usability for seniors and caregivers.
What Is CR Packaging for Blister Products?
Child-Resistant (CR) packaging for blister products is a secondary packaging solution designed to limit a child’s ability to access individual doses while still allowing adults to retrieve medication when needed.
In practice, this involves enclosing a blister card inside a protective carton or sleeve with built-in locking mechanisms or opening sequences. To be considered CR, the package must pass specific protocol testing under 16 CFR §1700.20, the standard set by the U.S. Consumer Product Safety Commission (CPSC).

CR Must Also Be Senior-Friendly (SF)
Under U.S. regulations, a package cannot be labeled Child-Resistant unless it is also proven to be Senior-Friendly (SF). That means:
CR/SF packaging ensures that safety doesn’t come at the expense of accessibility—an essential requirement for elderly patients and caregivers managing chronic medications.
FAQ: Child-Resistant / Senior-Friendly (CR/SF) Blister Packaging for Prescription Medications
CR/SF Certified Packaging Solutions from Keystone
Why Keystone?
Get in touch today to learn more about integrating CR/SF packaging into your pharmaceutical supply chain. Let Keystone Folding Box Co. help you meet regulatory demands while improving patient safety and satisfaction.